Abstract
Background: With an ever-growing need for efficiency in emergency medicine, especially at the collegiate level, there has been some debate about the necessity of carrying large machines such as the LIFEPAK to calls involving “non-critical” patients. Despite the numerous applications of the LIFEPAK – for example, cardiac monitoring and taking automatic blood pressure readings at precise time intervals – its price may be beyond the budget of many collegiate EMS crews, and its weight can make it a burden to haul, slowing response times in emergent situations. This potential decrease in efficiency may not be necessary, especially if the LIFEPAK is only used to obtain patient vitals, which crews are trained to obtain manually. Objectives: The purpose of this study was to answer the following question: do manual blood pressure and heart rate measurements differ substantially from those obtained using the LIFEPAK? By answering this question, it may be possible to evaluate whether the LIFEPAK is a worthwhile investment for collegiate EMS crews who don’t often use its more advanced features. Methods: Crews measured heart rates and blood pressures of student volunteers manually and using the LIFEPAK 15 device. These student volunteers were assumed to be proxies for patients meeting the criteria for a “non-critical” designation, made when a patient does not meet University Emergency Medical Response upgrade criteria for the Richardson Fire Department’s Advanced Life Support (ALS) Unit. Crews were asked to document blood pressure and heart rate readings using each method on a designated form. Results: It was concluded that there was no significant difference between the manual vitals and LIFEPAK vitals obtained by crews throughout the duration of this study. Therefore, manual vitals can be said to be a numerically consistent alternative to those taken using the LIFEPAK. Conclusions: As a result, we recommend that collegiate EMS crews conduct further cost-benefit analyses to assess if this substitution can improve the efficiency, response time, and satisfaction of crews, while reducing their physical burden.
Introduction
Campus-based emergency medical services (CBEMS) are becoming increasingly common as universities recognize the value of timely emergency responses on collegiate campuses. However, one of the challenges that these organizations often encounter is attempting to maximize crew efficiency while operating within the financial constraints of their annual budgets.
The LIFEPAK 15 and similar devices are utilized by emergency medical services (EMS) nationwide for their all-in-one advanced cardiac monitoring, defibrillation, and vital sign assessment capabilities (Stryker, 2009)1. By automating blood pressure and heart rate measurements, providers are instead able to focus more on administering treatments and ensuring patient comfort throughout their calls. Despite these advantages, however, the LIFEPAK 15’s size and weight may make it a burden for crews to transport, and its price may present a significant financial challenge for organizations on strict annual budgets.
University Emergency Medical Response (UEMR) at The University of Texas at Dallas is a 24/7 BLS (Basic Life Support) QRS (Quick Response Service) volunteer collegiate EMS agency that actively serves a population of around 30,000 people, including students, faculty, and campus visitors. In 2024 alone, UEMR crews, typically composed of two providers in a golf cart and one field captain in an SUV, provided care for a total of 394 patients. The standard equipment load of UEMR crews includes the LIFEPAK, in addition to two large bags, each containing equipment such as medical and trauma kits, oxygen tanks, and oxygen delivery devices.
Crews are required to bring the LIFEPAK device to every call which, at 18.5 lbs., may be cumbersome for them to transport along with all this essential medical equipment. For example, when attempting to reach a patient on the upper floor of a building without elevator access, crews may quickly fatigue from their nearly-50 lb. load, which might negatively impact their performance and efficiency while caring for a patient, though future studies would need to be conducted to determine this effect with certainty. Furthermore, in other collegiate EMS agencies where a full set of equipment may not be mandated, the added weight of the LIFEPAK and similar devices could lead crews to leave behind other equipment, which could negatively impact patient care if essential items are omitted.
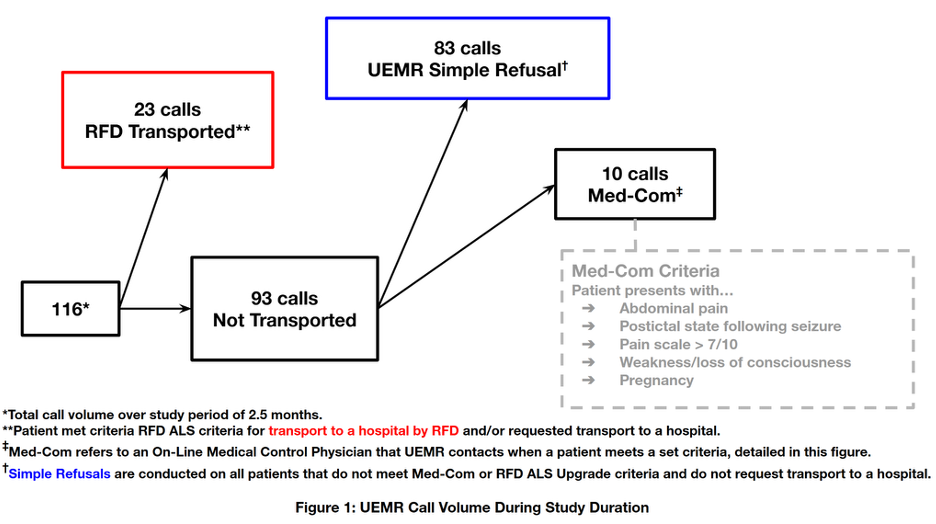
Approximately 71% of CBEMS report an annual budget of under $30,000: though most of these funds come from the institutions themselves, the rest must be secured from external sources beyond the host university2. The LIFEPAK 15 and other similarly functioning devices can frequently cost up to $48,000, meaning they can consume well over a CBEMS’s entire yearly budget3. This can leave the organization with insufficient funds to sustain continued provider education, equipment maintenance, transportation, and operational sustainability, especially for newly established CBEMS.
This study provides an analysis of the numerical differences between device and manual vital sign measurements to determine if a substitution of the latter would significantly affect patient care and treatment outcomes. The goal of this project is to assess if manual vitals may be an accurate, effective, and appropriate alternative for collegiate EMS crews seeking to improve their efficiency or re-prioritize their budget.
Methods
Inclusion and Exclusion Criteria
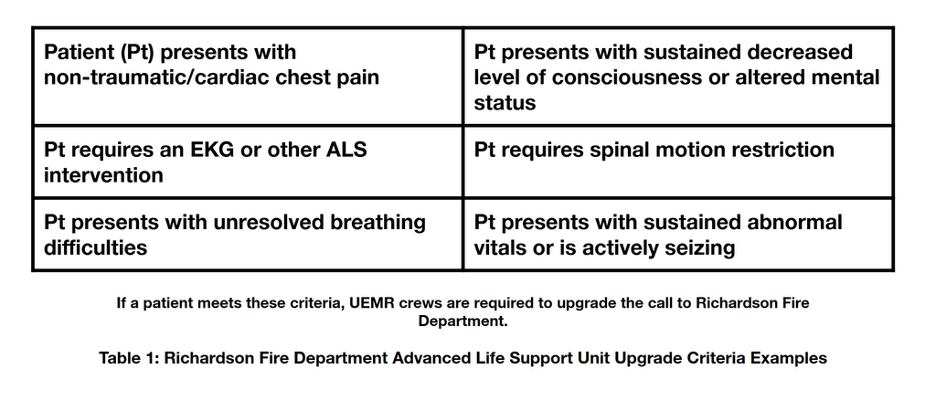
Blood pressure and heart rate measurements were obtained from student volunteers that served as proxies for patients meeting criteria for a “non-critical” designation. This was done in order to avoid any potential risk to patients as presentation and “non-critical” status may change throughout a medical emergency. “Non-critical” patients are operationally defined as patients who consent to treatment by UEMR and do not meet the Richardson Fire Department’s (RFD) Advanced Life Support (ALS) upgrade criteria (Table 1).
Seventy-nine student volunteers that consented to participation and met the above inclusion criteria were selected at locations around campus that UEMR crews are commonly dispatched to. This was done in an attempt to mirror the true number of non-critical patients and environments that UEMR would be dispatched to throughout the study duration (true call volume from the study duration is outlined in Figure 1).
Manual blood pressures were measured with a sphygmomanometer placed on bare skin. Crews alternated manual and LIFEPAK blood pressure measurements on the same arm at five-minute intervals. The measurement process proceeded as follows: crews took the first manual blood pressure measurement, then the first automated measurement five minutes later, and so on. All blood pressures were measured at the brachial artery. These measurements were obtained on consecutive volunteers with verbal consent obtained before each reading. This numerical data was collected and submitted to a Google Form that included a section where crews could note unique environmental conditions, device failures, user errors, and other disturbances or distractions that might have influenced the readings.
One patient’s measurements were excluded due to reported extreme background commotion that could have influenced the accuracy of the crew’s manual blood pressure readings. Additionally, seven volunteers’ heart rate measurements were excluded due to malfunctions of the LIFEPAK device: for example, a faulty connection between the pulse oximeter and the LIFEPAK that rendered an “error” reading for automatic heart rate measurements.
This study was approved by our Institutional Review Board under Exemption Category 2(i) as no patient records were accessed, the study posed no risk to student volunteers, and no identifiable personal information was recorded.
Data Collection
All measurements were first obtained manually, followed by corresponding readings using the LIFEPAK 15. Manual heart rates were assessed as a radial pulse. Blood pressures were collected using the method described previously.
Both manual measurements and those recorded using the LIFEPAK 15 were collected using a Google Form that crews completed while collecting this data from student volunteers. The results were subsequently tabled in a Google Sheets spreadsheet. The timestamp column recorded by the Google Form upon submission was deleted and the data rows were shuffled weekly using randomized sort functions in Google Sheets to avoid potential bias during analysis. Upon the conclusion of data collection, each data set was assigned a unique Call ID letter for ease of comparison.
Numerical differences between vitals obtained manually and using the LIFEPAK device were calculated and assessed. All of the data obtained followed a normal distribution, allowing for statistical analysis using a paired-sample t-test to assess potential differences between the two measurement methods.
Data Analysis
A threshold of p < 0.05 was established for a broad examination of the significance of the numerical differences between systolic blood pressure (SBP), diastolic blood pressure (DBP), and heart rates (HR) obtained manually and with the LIFEPAK 15. As mentioned, a paired t-test was conducted using Google Sheets once the data was revealed to exhibit normal behavior. Further analysis was conducted by evaluating the differences between manually obtained vitals and LIFEPAK 15 measurements within both the 95% confidence interval and a novelly defined clinical significance range of ±10 mmHg for blood pressure measurements and ±10 bpm for heart rate measurements.
Establishing the Clinical Significance Range for Blood Pressure and Heart Rate Measurements
UEMR crews define a consistent blood pressure as one that varies less than 10 mmHg and a consistent heart rate as one that varies less than 10 to 15 bpm between measurements throughout the duration of a call, taking into account variation due to patient stress levels and incident severity. Variation beyond this may dictate varying courses of treatment and/or necessitate contacting our on-line medical control or upgrading our patient for transport to a hospital by RFD. Additionally, blood pressure ranges are defined in 10 mmHg intervals by the Cleveland Clinic4, with each range indicating a different level of risk for conditions such as cardiovascular disease, stroke, and aneurysms.
Since these ranges of ±10 mmHg and ±10 bpm also fell within the limits of our calculated 95% confidence intervals, and appeared to be the most restrictive boundaries for what is considered a consistent, these were established as our “boundaries of clinical significance” for differences in blood pressure and heart rate measurements.
A Bland-Altman plot was selected to display this data as well as the two intervals chosen to highlight significance or lack thereof5.
Results
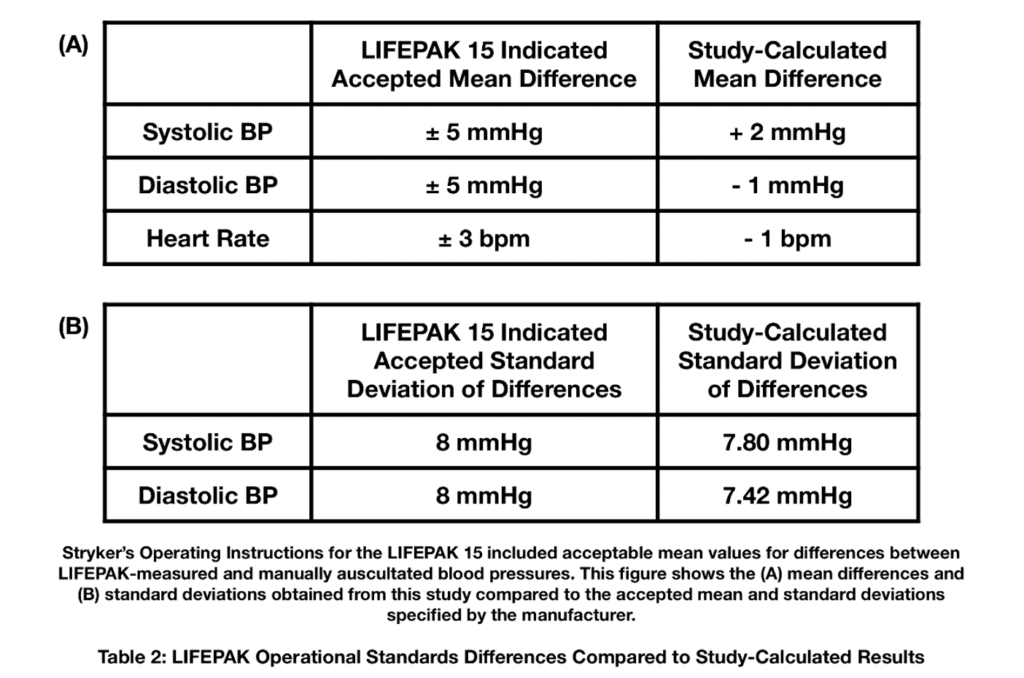
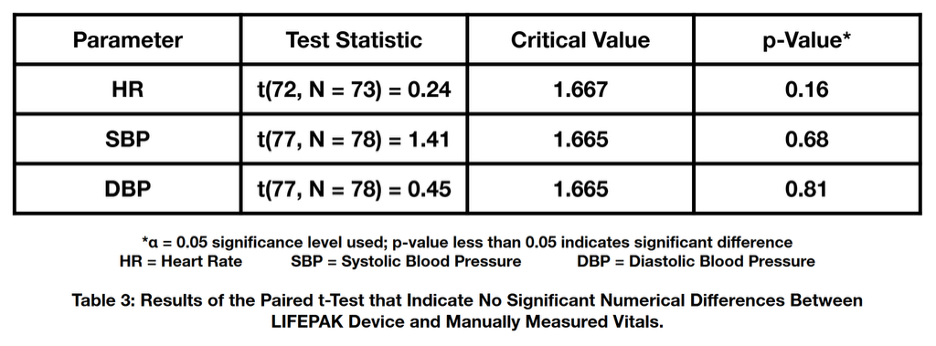
The LIFEPAK 15 Operating Instructions6 provided by Stryker indicate that an acceptable mean difference between non-invasive blood pressure (NIBP) readings and auscultatory readings was ±5 mmHg with a standard deviation of 8 mmHg (corresponding guidelines for differences in automated and manual heart rate measurements was ±3 bpm). Thus, as indicated by the results in Table 2, it was concluded that there was no significant difference between the numerical values obtained using each method. The specific results of the paired t-test are shown in Table 3. These findings provide evidence that manual methods produced measurements comparable to the readings from the LIFEPAK device for these parameters.
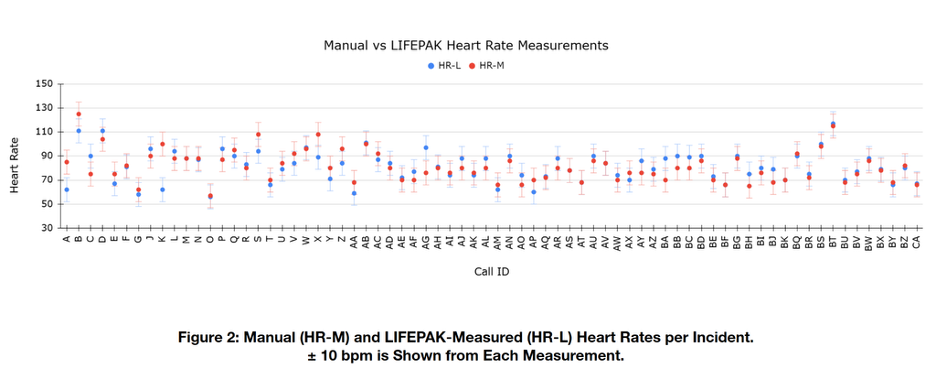
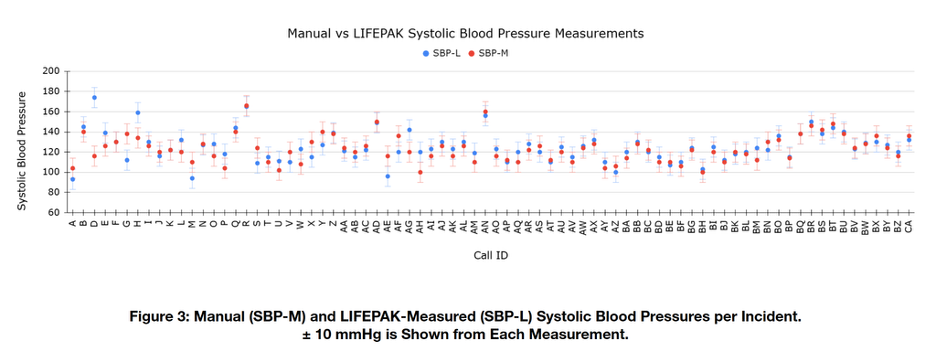
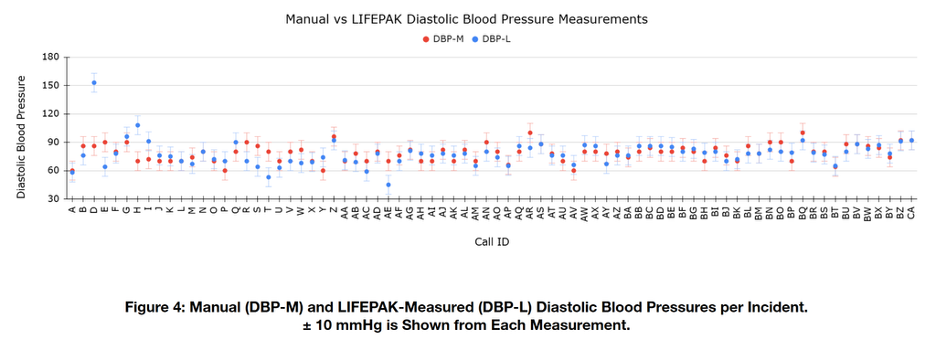
Upon analysis of the calculated 95% confidence interval, it was observed that blood pressure differences of up to approximately 20 mmHg remained within the interval’s boundaries. These points were outside the range of what UEMR classifies as consistent, however, they would not be considered to display significant differences from the confidence interval alone. This necessitated the establishment of the novel boundaries, described previously, as the threshold for “Clinically Significant Differences.” As mentioned, this interval was chosen based on the assumption that differences of 10 mmHg or greater may result in differences to treatment strategies, as well as increases in levels of risk for adverse health outcomes7,8,9,10.
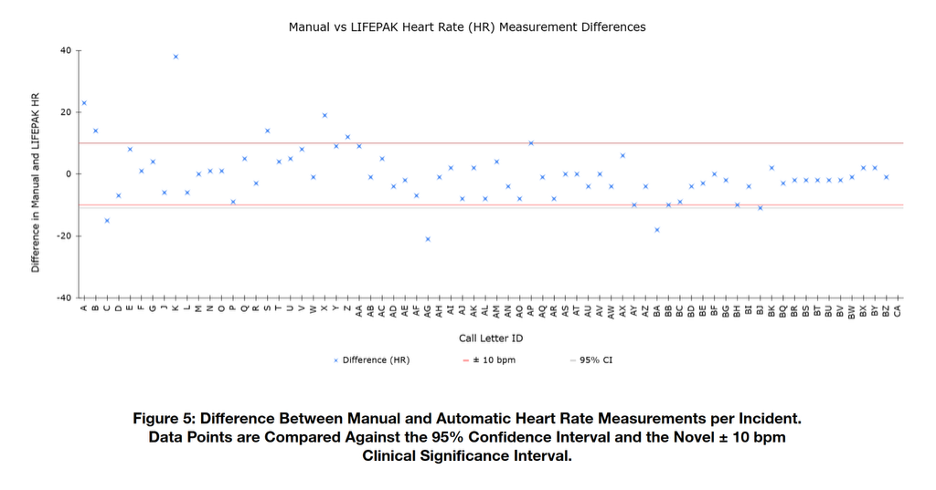
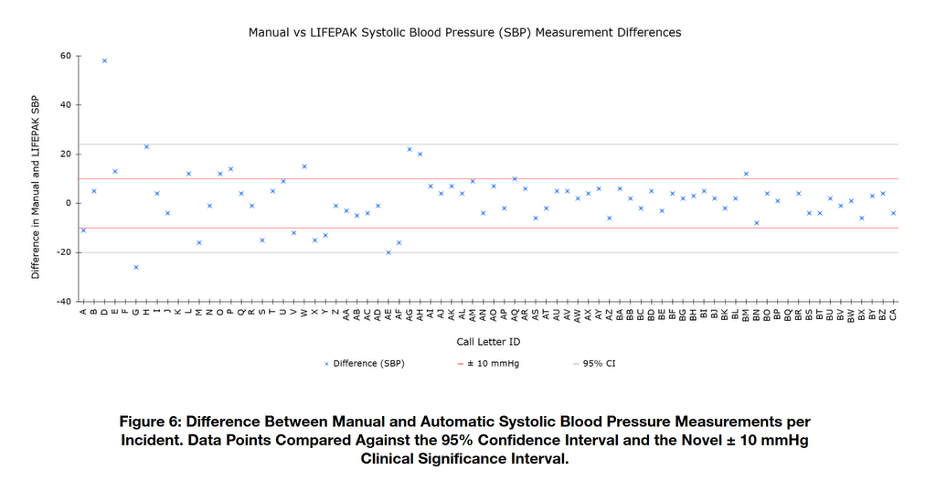
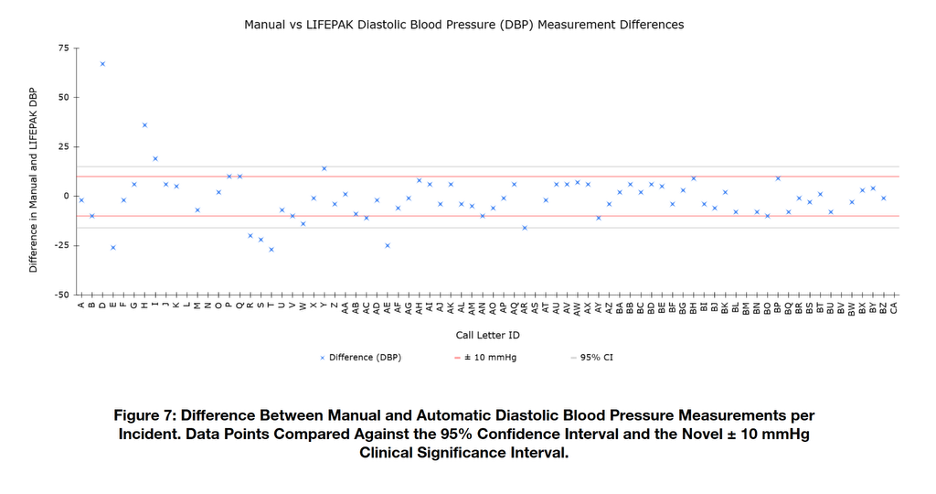
Our results indicate that there is no statistically significant difference between the two methods of vital sign monitoring. In addition, it was determined that most differences across all parameters measured fell within the clinically significant boundaries of ±10 mmHg and ±10 bpm for blood pressures and heart rates, respectively. These results can be seen in Table 4.
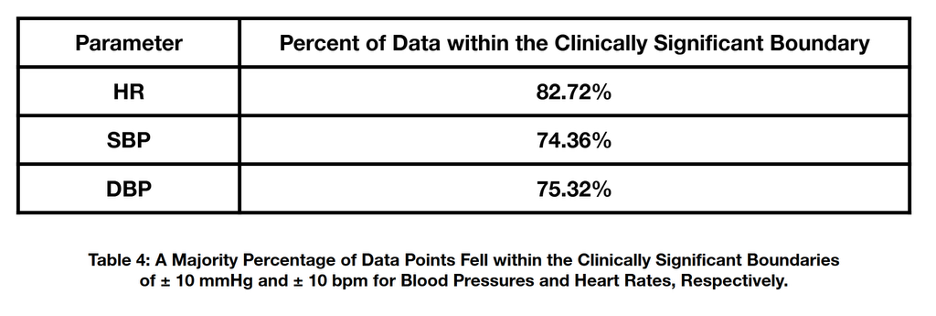
It is apparent that most of our data demonstrated no clinically significant numerical differences in blood pressure and heart rate measurements between the two methods of vital sign monitoring. These findings can be extrapolated to suggest that, for most of our non-critical, non-transport patients, the method of monitoring vital signs used would not impact the treatment protocols employed.
Discussion
CBEMS are a relatively recent addition to the EMS community with the first CBEMS organization established in 1968 at Cedarville University11. These organizations differ significantly in their operational scope: for example, in whether they function at the basic life support (BLS) or advanced life support (ALS) level, whether they transport patients directly to a local hospital rather than treating patients on-site, and the extent of financial support they receive from their respective institutions.
Notably, CBEMS that are required to secure external funding from sources outside their universities may face financial challenges including those involved in maintaining a stable or consistent annual budget that affect the organization’s ability to properly allocate its resources and preserve long-term stability. Given this, it is critical for CBEMS to adopt carefully structured financial plans that prioritize the allocation of sufficient resources to maintain and repair essential crew equipment, while ensuring that funds are sustained for ongoing provider education and training to maintain appropriate and efficient crew responses to calls.
Our analysis provides compelling evidence that there is no clinically significant numerical difference between vitals measured using the LIFEPAK and those obtained manually. For agencies such as UEMR that operate in a non-transporting and BLS capacity, we conclude that manual methods of vital sign monitoring are an acceptable, accurate, and reliable alternative to the LIFEPAK.
This substitution may be particularly beneficial for non-transporting BLS agencies that do not typically utilize the LIFEPAK’s advanced functionalities such as cardiac monitoring and capnography. For CBEMS agencies that prefer automated vital sign monitoring or those that require functions beyond basic vital sign monitoring such as cardiac monitoring, capnography, and 12-lead capabilities, more cost-effective alternatives to the LIFEPAK also exist: external electrocardiogram (EKG) machines are available at a fraction of both the cost and weight of the LIFEPAK, for example. However, further studies are necessary to determine if such external EKG machines are appropriate replacements both in terms of accuracy and efficiency.
For established agencies that are financially stable and/or those that already own a LIFEPAK or similar device, there are still substantial benefits to reconsidering its use and favoring smaller, handheld devices such as a pulse oximeter and sphygmomanometer. Not only does this substitution significantly reduce the weight of equipment that crews must carry to patients, but it may also allow for an enhancement of crew mobility, potentially improving operational efficiency, crew performance, and response times, though further research is necessary to determine whether this improvement is significant. Additionally, encouraging providers to manually measure and monitor vital signs may enhance clinical skills and promote provider accountability12,13.
Despite these benefits, the substitution of manual for automated monitoring may reduce provider satisfaction as the convenience of gathering and transmitting measured vital signs to other agencies or hospitals is lost14.
It should be noted that this study assumes equal provider competency across non-transporting BLS agencies in the United States, and results should not be extrapolated to ALS or transporting agencies. Furthermore, the results of this study are applicable only to the LIFEPAK 15: though all FDA-approved automated vital sign monitoring devices must meet industry standards and should be expected to result in relatively consistent measurements, it is possible that older, refurbished editions may have sustained slight calibration shifts that could produce variation by a few units. Further studies using other devices would be needed to ascertain the feasibility of manual vital signs monitoring as an alternative for these.
Furthermore, the population for most CBEMS is largely younger, generally healthy patients; therefore, the results of this study are not generalizable outside of this population. Vital signs beyond blood pressure and heart rate were not addressed.
It should be noted that studies have indicated that discrepancies between manual and automated blood pressure measurements may be amplified in patients admitted to intensive care and other specialty units15. Therefore, further studies are required to assess whether this conclusion is maintained in patients designated as critical.
Because the designation of a patient as “non-critical” is made throughout the duration of a call, it is important to note that devices such as AEDs that are utilized at the BLS level should still be carried by crews, even if a full ALS monitor is not required for the agency’s scope of practice.
Conclusion
This study examined the discrepancies between manual and automated blood pressure and heart rate readings, revealing no significant difference between the two methods. Additionally, no clinically significant differences between the two methods of vital sign monitoring were observed. Therefore, we conclude that manually auscultated blood pressures and radial heart rates are viable alternatives for automatic, LIFEPAK device-measured NIBPs and heart rates. As a result, we recommend other CBEMS operating at a BLS-QRS level evaluate the feasibility of this substitution for their agency’s particular clinical and operational needs. Further studies will be conducted to ascertain if this substitution results in a difference in crew efficiency and/or satisfaction.
Acknowledgements
The authors would like to thank the University of Texas at Dallas community for their participation in this study, as well as Sheila Elliot and Nick Spray for their continued support.
References
1. Deathcheater. National EMS Museum. 2014
2. Bartho MJ Determinants of Volunteer Collegiate-Based Emergency Medical Service Budget Size. JCEMS. 2021.
3. AED Professionals. Physio-control/stryker lifepak 15 defibrillator.
4. Professional CC medical. What is Blood Pressure? Cleveland Clinic. 2022.
5. Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1(8476):307-310.
6. Physio-Control. LIFEPAK 15 monitor/defibrillator – Operating instructions. Physio-Control, Inc.. 2019.
7. Clark CE, Taylor RS, Shore AC, Campbell JL. The difference in blood pressure readings between arms and survival: primary care cohort study. BMJ. 2012;344:e1327. Published 2012 Mar 20. doi:10.1136/bmj.e1327
8. Philip R, Beaney T, Appelbaum N, et al. Variation in hypertension clinical practice guidelines: a global comparison. BMC Med. 2021;19(1):117. Published 2021 May 12. doi:10.1186/s12916-021-01963-0
9. Ettehad D, Emdin CA, Kiran A, et al. Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet. 2016;387(10022):957-967. doi:10.1016/S0140-6736(15)01225-8
10. Mancia G, Kreutz R, Brunström M, et al. 2023 ESH Guidelines for the management of arterial hypertension The Task Force for the management of arterial hypertension of the European Society of Hypertension: Endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA) [published correction appears in J Hypertens. 2024 Jan 1;42(1):194. doi: 10.1097/HJH.0000000000003621.]. J Hypertens. 2023;41(12):1874-2071. doi:10.1097/HJH.0000000000003480
11. Kirychuk N. America’s First Collegiate Emergency Medical Service Turns 50. Cedarville University News. 2018.
12. Areia C, King E, Ede J, et al. Experiences of current vital signs monitoring practices and views of wearable monitoring: A qualitative study in patients and nurses. J Adv Nurs. 2022;78(3):810-822. doi:10.1111/jan.15055
13. Rock M. Ditch the Machine to Improve Accuracy in Blood Pressure Measurement and Diagnostics. JEMS. 2018
14. Wood J, Finkelstein J. Comparison of Automated and Manual Vital Sign Collection at Hospital Wards. Stud Health Technol Inform. 2013;190:48-50.
15. Mirdamadi A, Etebari M. Comparison of manual versus automated blood pressure measurement in intensive care unit, coronary care unit, and emergency room. ARYA Atheroscler. 2017;13(1):29-34.
Author & Article Information
Priya Darbha, EMT-B, is a volunteer EMT with University of Texas at Dallas University Emergency Medical Response and serves as the current Research Lieutenant for the organization. Hitankshini Pranav Pandya, BS Neuroscience, EMT-B, is a volunteer EMT with University of Texas at Dallas University Emergency Medical Response and is the former Research Lieutenant for the organization. Tisha Smitha Gautam, BS Biology, EMT-B, is a volunteer EMT with University of Texas at Dallas University Emergency Medical Response and is the former Administrative Chief for the organization. Divya Arivalagan, EMT-B, is a volunteer EMT with University of Texas at Dallas University Emergency Medical Response and serves as the current Administrative Chief for the organization.
Author Affiliations: From University Emergency Medical Response – The University of Texas at Dallas – Richardson, TX (P.D., H.P., T.G., D.A.)
Address for Correspondence: Priya Darbha, EMT-B | Email: priya.darbha@utdallas.edu
Conflicts of Interest/Funding Sources: By the JCEMS Submission Declaration Form, all authors are required to disclose all potential conflicts of interest and funding sources. All authors declared no conflicts of interest
Authorship Criteria: By the JCEMS Submission Declaration Form, all authors are required to attest to meeting the four ICMJE.org authorship criteria: (1) Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; AND (2) Drafting the work or revising it critically for important intellectual content; AND (3) Final approval of the version to be published; AND (4) Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Submission History: Received March 28, 2025, accepted for publication June 23, 2025
Published Online: September 06, 2025
Published in Print: Pending
Reviewer Information: In accordance with JCEMS editorial policy, Original Research manuscripts undergo double-blind peer review by at least two independent reviewers. JCEMS thanks the anonymous reviewers who contributed to the review of this work.
Copyright: © 2025 Darbha, Pandya, Gautam, & Arivalagan. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. The full license is available at: https://creativecommons.org/licenses/by/4.0/
Electronic Link: https://doi.org/10.30542/JCEMS.2026.08.01.10
Photo Credits: Photo courtesy of the authorship team.





